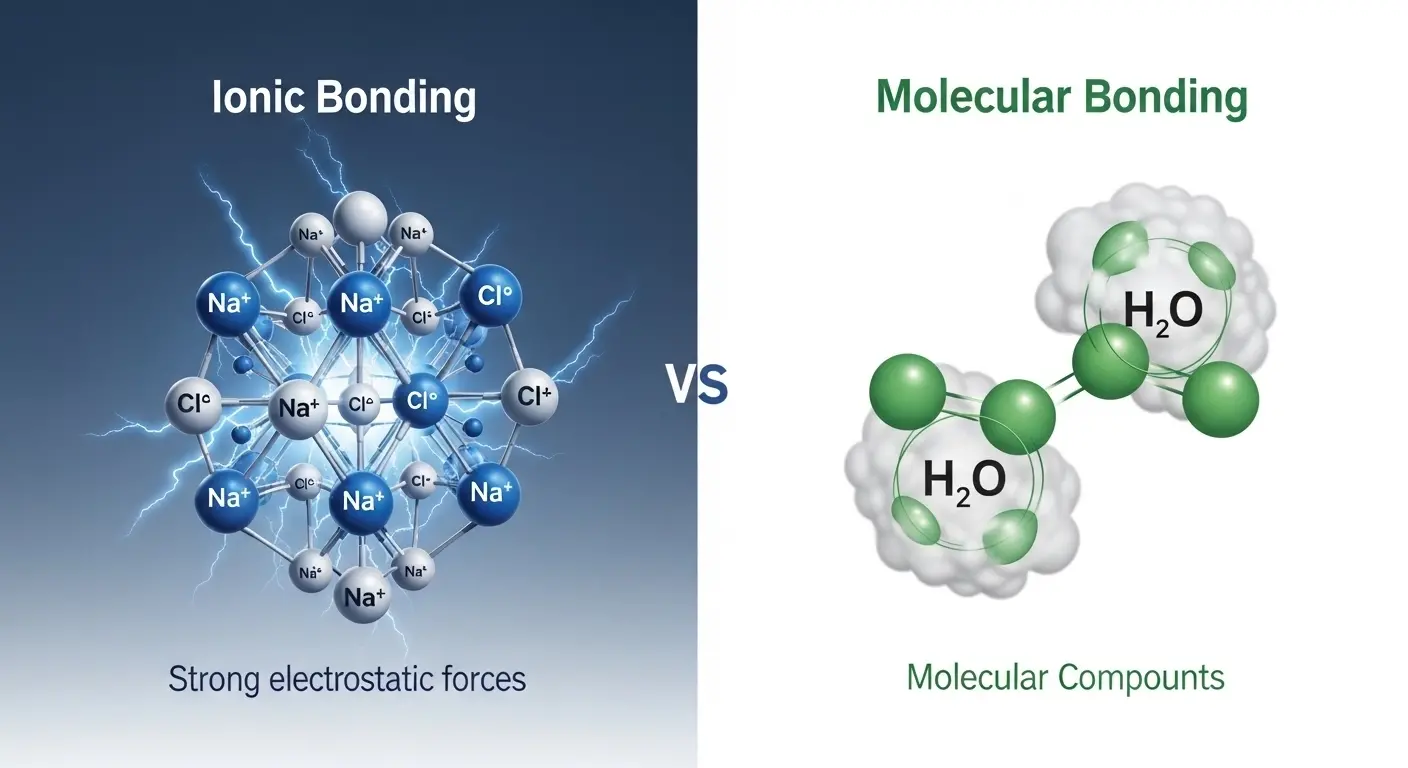
The difference between ionic and molecular substances often confuses students when they first step into chemistry. Imagine a classroom where salt crystals are passed around during an experiment, and at the same time, students smell vinegar or perfume.
One feels solid and structured, while the other seems to vanish into the air. This everyday contrast is actually the foundation of the difference between ionic and molecular compounds.
When we talk about the difference between ionic and molecular, we are really exploring how atoms choose to behave either by transferring electrons or sharing them. The difference between ionic and molecular is not just a chemistry topic; it explains why salt dissolves in water but sugar behaves differently in heat.
Understanding the difference between ionic and molecular helps us connect science with real-life observations in food, medicine, and nature.
Pronunciation
- Ionic
- US: /aɪˈɑːnɪk/
- UK: /aɪˈɒnɪk/
- Molecular
- US: /məˈlekjələr/
- UK: /məˈlekjʊlə/
Key Difference Between Ionic and Molecular
Before diving deeper, remember this simple idea:
- Ionic = transfer of electrons (forms charged ions)
- Molecular = sharing of electrons (forms neutral molecules)
This basic contrast leads to many physical and chemical differences that we will explore below.
Difference Between Stout and Porter: in (2026)
Why Understanding Ionic and Molecular Differences is Important
Knowing the difference between ionic and molecular compounds is essential for students, scientists, doctors, and even industries. In medicine, ionic compounds help create electrolytes in our body, while molecular compounds form many organic drugs. In agriculture, fertilizers depend on ionic compounds, whereas pesticides often rely on molecular structures.
Without understanding the difference between ionic and molecular, it becomes difficult to explain why some substances conduct electricity while others do not. This knowledge also supports innovation in technology, environmental science, and food chemistry. In society, it helps us use materials safely and effectively in daily life.
Difference Between Choose and Chose: in (2026)
Transition to Main Discussion
Now that we understand the basic idea and importance, let’s explore the difference between ionic and molecular compounds in detail.
Difference Between Ionic and Molecular
1. Formation of Bond
- Ionic: Formed by transfer of electrons
- Example 1: NaCl (sodium chloride)
- Example 2: MgO (magnesium oxide)
- Molecular: Formed by sharing electrons
- Example 1: H₂O (water)
- Example 2: CO₂ (carbon dioxide)
2. Type of Particles
- Ionic: Made of ions (positive and negative)
- Example 1: Na⁺ and Cl⁻ in salt
- Example 2: Ca²⁺ and O²⁻ in calcium oxide
- Molecular: Made of molecules
- Example 1: O₂ (oxygen gas)
- Example 2: CH₄ (methane)
3. Electrical Conductivity
- Ionic: Conduct electricity in molten or aqueous state
- Example 1: Molten NaCl conducts electricity
- Example 2: Saltwater solution conducts current
- Molecular: Do not conduct electricity
- Example 1: Sugar solution does not conduct
- Example 2: Alcohol solution does not conduct
4. Melting and Boiling Point
- Ionic: High melting and boiling points
- Example 1: NaCl melts at very high temperature
- Example 2: MgO requires extreme heat
- Molecular: Low melting and boiling points
- Example 1: Ice melts easily
- Example 2: CO₂ turns into gas quickly
5. Physical State
- Ionic: Usually solid crystals
- Example 1: Table salt
- Example 2: Potassium chloride
- Molecular: Can be gas, liquid, or soft solid
- Example 1: Oxygen (gas)
- Example 2: Water (liquid)
6. Solubility
- Ionic: Usually soluble in water
- Example 1: NaCl dissolves in water
- Example 2: KBr dissolves in water
- Molecular: Variable solubility
- Example 1: Sugar dissolves in water
- Example 2: Oil does not dissolve
7. Structure
- Ionic: Giant lattice structure
- Example 1: Sodium chloride crystal
- Example 2: Calcium fluoride lattice
- Molecular: Individual molecules
- Example 1: Water molecules
- Example 2: Methane molecules
8. Hardness
- Ionic: Hard but brittle
- Example 1: Salt crystals break easily
- Example 2: Rock-like ionic compounds
- Molecular: Soft or flexible
- Example 1: Wax
- Example 2: Sulfur (molecular form)
9. Bond Strength
- Ionic: Strong electrostatic forces
- Example 1: NaCl bond strength
- Example 2: MgO strong attraction
- Molecular: Weaker intermolecular forces
- Example 1: Water hydrogen bonds
- Example 2: CO₂ weak attraction
10. Electrical Charge
- Ionic: Carry full charges
- Example 1: Na⁺ ion
- Example 2: Cl⁻ ion
- Molecular: Neutral overall
- Example 1: H₂O neutral molecule
- Example 2: CO₂ neutral molecule
Nature and Behaviour
Ionic compounds behave like tightly packed structures that need high energy to break. They are stable, rigid, and form crystals. Molecular compounds behave more flexibly, often existing as gases or liquids with smoother interactions. They are more reactive in biological systems because they can easily interact through shared electrons.
Why People Get Confused
People often confuse ionic and molecular compounds because both involve atoms bonding together. Additionally, some compounds show mixed properties, making classification difficult. For example, substances like hydrogen chloride behave differently in gas and aqueous form, increasing confusion among learners.
Table: Difference and Similarity
| Feature | Ionic | Molecular |
| Bond Type | Electron transfer | Electron sharing |
| Structure | Crystal lattice | Individual molecules |
| Conductivity | Conducts in solution/molten | Does not conduct |
| Melting Point | High | Low |
| Charge | Charged ions | Neutral |
| Solubility | Usually soluble in water | Variable |
| Physical State | Solid | Gas/liquid/solid |
| Strength | Strong bonds | Weaker forces |
Similarity: Both are formed from atoms and follow chemical bonding principles.
Which is Better in What Situation?
Ionic compounds are more suitable in situations requiring strength, stability, and electrical conductivity, such as batteries, salt-based solutions, and industrial materials. Their ability to dissociate into ions makes them essential in biological processes like nerve signaling.
Molecular compounds are better suited for biological systems, fuels, and gases because of their flexibility and lower reactivity. Water, oxygen, and glucose are molecular compounds that sustain life. Therefore, neither is “better” overall; their usefulness depends entirely on the context of application.
Metaphors and Similes
- Ionic compounds are like “a strong handshake that cannot be easily broken.”
- Molecular compounds are like “friends sharing secrets instead of exchanging ownership.”
Connotative Meaning
- Ionic: Strong, rigid, stable (positive connotation in science, but can feel harsh in structure)
- Example: “Salt crystals are ionic bonds holding tightly together.”
- Molecular: Flexible, soft, dynamic (positive and neutral connotation)
- Example: “Water is a molecular substance flowing freely.”
Idioms or Proverbs
- “Like salt in water” – used to describe complete dissolution (ionic behavior example)
- “Two molecules in harmony” – used metaphorically for close relationships
Works in Literature
- The Disappearing Spoon – Sam Kean (Science narrative, 2010)
- Molecules of Emotion – Candace Pert (Science/psychology, 1997)
- The Chemical History of a Candle – Michael Faraday (Science lectures, 1861)
Movies Related to Chemistry Concepts
- Osmosis Jones (2001, USA) – Biological molecules inside the body
- The Martian (2015, USA) – Chemical and molecular survival science
- Breaking Bad (2008–2013, USA) – Chemistry-based molecular reactions
Frequently Asked Questions
1. What is the main difference between ionic and molecular compounds?
Ionic compounds transfer electrons, while molecular compounds share electrons.
2. Why do ionic compounds conduct electricity?
Because they form ions that move freely in liquid or molten state.
3. Are molecular compounds always gases?
No, they can be gases, liquids, or solids.
4. Which is stronger, ionic or molecular bonds?
Ionic bonds are generally stronger due to electrostatic attraction.
5. Can a compound be both ionic and molecular?
Some compounds show mixed behavior depending on conditions.
How Both Are Useful in Surroundings
Ionic compounds are vital in soil fertility, water balance, and electrical systems. Molecular compounds support respiration, food chemistry, and energy cycles. Together, they maintain environmental balance and biological functions on Earth.
Final Words
Both ionic and molecular compounds are essential building blocks of chemistry. They may differ in structure, behavior, and properties, but together they explain how matter functions in the universe. From the salt in our food to the oxygen we breathe, both types of bonding shape life as we know it.
Conclusion
Understanding the difference between ionic and molecular compounds helps us connect microscopic chemical behavior with real-world applications. Ionic compounds show strength, stability, and conductivity, while molecular compounds offer flexibility and diversity in nature.
This distinction is crucial in education, medicine, agriculture, and industry. By learning how atoms interact through ionic or molecular bonding, we gain a deeper appreciation of how everything around us is formed. Ultimately, the difference between ionic and molecular is not just scientific—it is a key to understanding life itself.

SwiftHarbor is a dedicated English professor, language researcher, and the founder of SpellCompare.com. With years of academic experience in English grammar, vocabulary development, and linguistic comparison, SwiftHarbor specializes in simplifying complex language rules into clear, practical explanations.
As an expert in word usage, spelling differences, and commonly confused terms, SwiftHarbor has helped thousands of learners improve their writing accuracy and communication skills. Through SpellCompare.com, he provides detailed comparisons, easy examples, and research-based insights that make English learning accessible for students, writers, and professionals worldwide.
Known for a clear teaching style and structured explanations, SwiftHarbor focuses on eliminating confusion between similar words, improving grammar confidence, and promoting precise communication. His mission is simple: to make English easier, clearer, and more powerful for everyone.