Understanding the difference between mass and density is one of the most important steps in learning basic physics. Many students often mix these two concepts, but the difference between mass and density is actually simple when explained with real-life examples. In science and daily life, the difference between mass and density helps us understand why some objects are heavy, why some float, and how matter behaves in different conditions.
Imagine a teacher holding a small iron ball and a large cotton bag. The iron ball feels heavy even though it is small, while the cotton bag is large but light. This real-world example shows the basic idea behind mass and density. Students often get confused between them, but once the difference between mass and density becomes clear, physics becomes much easier and more interesting.
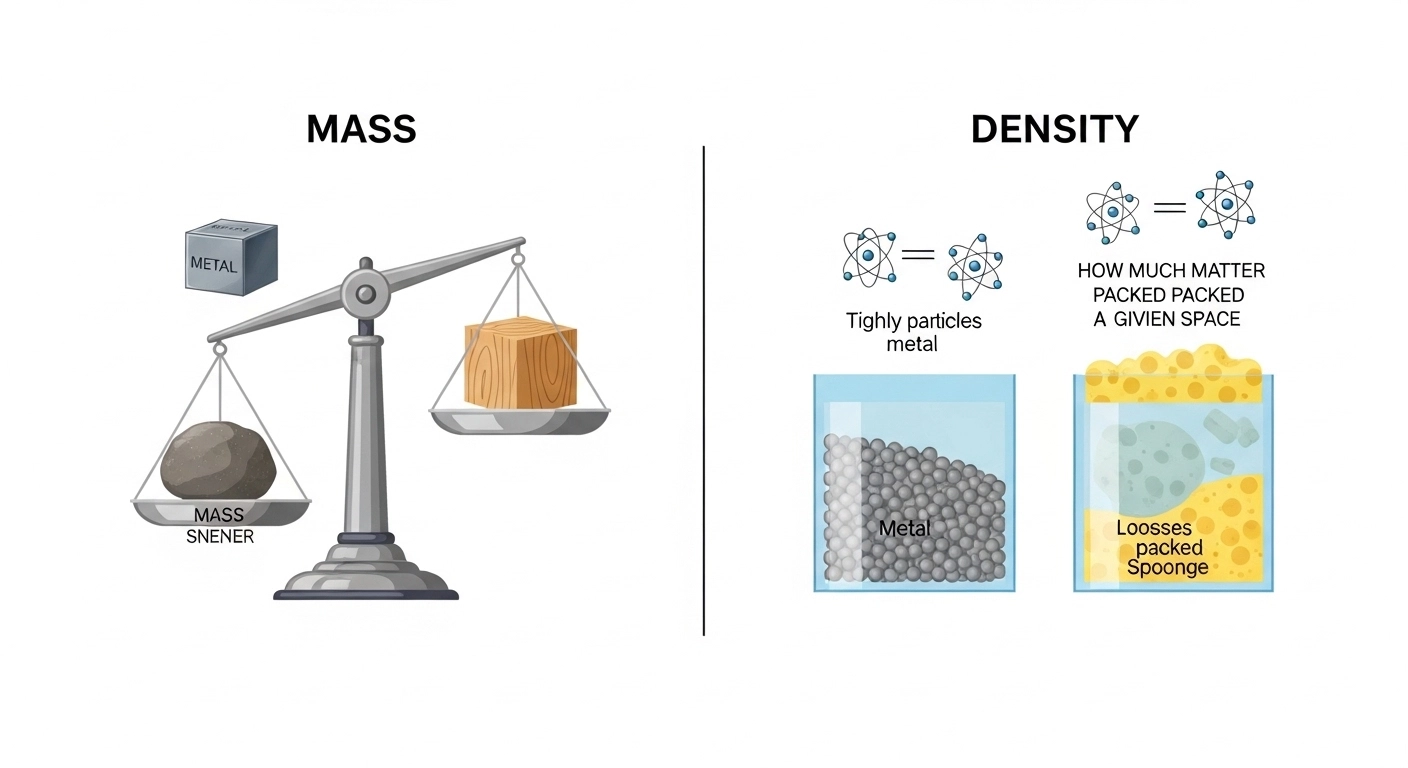
Mass tells us how much matter is in an object, while density tells us how tightly that matter is packed. Both are related, but they describe completely different properties of matter.
Key Difference Between Mass and Density
- Mass: Amount of matter in an object
- Density: Mass per unit volume of an object
Why Their Difference Matters in Learning and Society
Understanding the difference between mass and density is essential for students, engineers, scientists, and even doctors. In society, these concepts help in construction, transportation, medicine, and environmental studies. For example, engineers use density to choose materials for buildings, while scientists use mass to calculate force and motion.
If learners understand the difference between mass and density, they can better understand physical laws like gravity, buoyancy, and pressure. This knowledge is also important in industries such as aviation, shipbuilding, and manufacturing, where material properties determine safety and performance.
Pronunciation (US & UK)
- Mass
- US: /mæs/
- UK: /mæs/
- Density
- US: /ˈdɛn.sə.ti/
- UK: /ˈdɛn.sɪ.ti/
Linking Hook to Main Section
Now that you understand the basic idea of mass and density, let’s explore the difference between mass and density in detail through clear comparisons.
H2: Difference Between Mass and Density
1. Definition
- Mass: Amount of matter in an object
- Example 1: A rock has more mass than a ball.
- Example 2: A car has more mass than a bicycle.
- Density: How tightly matter is packed in a substance
- Example 1: Iron is denser than wood.
- Example 2: Water is denser than oil.
2. Symbol
- Mass: m
- Example 1: m = 10 kg
- Example 2: m = 5 kg
- Density: ρ (rho)
- Example 1: ρ = 1 g/cm³
- Example 2: ρ = 7.8 g/cm³ (iron)
3. Formula
- Mass: m = density × volume (indirectly used)
- Example 1: Calculating weight in physics problems
- Example 2: Determining object matter content
- Density: ρ = mass / volume
- Example 1: Finding material density
- Example 2: Comparing substances
4. SI Unit
- Mass: kilogram (kg)
- Example 1: 1 kg sugar
- Example 2: 50 kg person
- Density: kg/m³
- Example 1: Water = 1000 kg/m³
- Example 2: Air = 1.2 kg/m³
5. Dependence
- Mass: Independent of volume
- Example 1: Small iron piece still has mass
- Example 2: Big balloon has mass
- Density: Depends on both mass and volume
- Example 1: Same mass, different size → different density
- Example 2: Compressing gas increases density
6. Physical Property
- Mass: Scalar quantity
- Example 1: 10 kg (no direction)
- Example 2: 2 kg object
- Density: Derived physical property
- Example 1: Calculated value
- Example 2: Used in material science
7. Measurement Tool
- Mass: Weighing scale
- Example 1: Grocery scale
- Example 2: Laboratory balance
- Density: Calculated using instruments
- Example 1: Hydrometer
- Example 2: Pycnometer
8. Change Behavior
- Mass: Does not change easily
- Example 1: Object retains mass in space
- Example 2: Stone mass remains same
- Density: Can change with temperature/pressure
- Example 1: Air density changes in heat
- Example 2: Water density slightly changes with temperature
9. Role in Physics
- Mass: Used in force (F = ma)
- Example 1: Motion of car
- Example 2: Falling objects
- Density: Used in buoyancy
- Example 1: Ship floating
- Example 2: Ice floating on water
10. Nature of Quantity
- Mass: Fundamental quantity
- Example 1: Basic SI unit
- Example 2: Used in all mechanics
- Density: Derived quantity
- Example 1: Combination of mass & volume
- Example 2: Used in material comparison
Nature and Behaviour
- Mass Behaviour: Constant, stable, and intrinsic to matter. It does not depend on the environment.
- Density Behaviour: Variable, dynamic, and dependent on external conditions like pressure and temperature.
Why People Are Confused
People confuse mass and density because both involve matter and are often taught together. Also, both appear in similar formulas, making students think they are interchangeable. However, mass is “how much matter,” while density is “how compact that matter is.”
Table: Difference and Similarity Between Mass and Density
| Feature | Mass | Density |
| Definition | Amount of matter | Mass per unit volume |
| Symbol | m | ρ |
| Unit | kg | kg/m³ |
| Dependence | Independent | Depends on mass & volume |
| Type | Scalar | Derived |
| Nature | Constant | Variable |
| Measurement | Scale | Calculation |
| Use | Motion & force | Buoyancy & materials |
Which is Better in What Situation?
Mass (Better for Motion and Forces)
Mass is more important when studying motion, gravity, and force. For example, in physics problems involving acceleration or weight, mass is essential. Engineers use mass to calculate load-bearing capacity of structures. It is also important in space science, where mass determines gravitational pull and orbital motion. Without mass, basic mechanics cannot be studied. It is fundamental for understanding how objects move and interact in the physical world.
Density (Better for Materials and Fluids)
Density is more useful when studying materials, fluids, and buoyancy. For example, density helps determine why ships float and stones sink. It is widely used in chemistry, engineering, and environmental science. Density helps identify substances and compare materials. It is also important in weather forecasting and ocean studies. Without density, we cannot understand fluid behavior or material composition in real-world applications like construction and transportation.
Metaphors and Similes
- Mass is like the weight of a backpack full of books—it tells how much is inside.
- Density is like how tightly books are packed in the backpack.
Connotative Meaning
- Mass: Neutral (represents quantity of matter)
- Example: “This object has a large mass.”
- Density: Neutral to positive (represents structure and compactness)
- Example: “Gold has high density, making it valuable.”
Idioms or Proverbs
- “Heavy as lead” → refers to high mass objects
- Example: “The stone is heavy as lead.”
- “Packed like sardines” → relates to high density
- Example: “The room was packed like sardines.”
Works in Literature
- Principles of Physics – Resnick & Halliday (Science textbook, 1960s)
- The Feynman Lectures on Physics – Richard Feynman (Physics, 1964)
Movies Related to Science Concepts
- Interstellar (2014, USA)
- The Martian (2015, USA)
Frequently Asked Questions (FAQs)
1. Is mass the same as density?
No, mass is matter amount while density is mass per volume.
2. Can density change?
Yes, with temperature and pressure.
3. What is the SI unit of mass?
Kilogram (kg).
4. What is the SI unit of density?
kg/m³.
5. Why do objects float?
Because their density is less than the liquid.
How Both Are Useful for Surroundings
Mass helps in measuring objects in daily life like groceries and construction materials. Density helps in understanding natural phenomena like floating, sinking, and atmospheric conditions. Both together explain how matter behaves in nature.
Final Words
Mass and density are fundamental physical concepts that explain the behavior of matter in the universe. While mass tells us how much matter an object contains, density explains how that matter is arranged. Both are essential in science, engineering, and everyday life. Understanding them not only improves academic knowledge but also helps in real-world decision-making, from building structures to studying nature.
Conclusion
The difference between mass and density is a fundamental concept in physics that helps us understand matter in a deeper way. Mass represents the total amount of matter in an object, while density explains how that matter is distributed within a given space.
Although they are closely related, their functions and applications are completely different. Mass is used in studying motion, force, and gravity, whereas density is essential in material science, buoyancy, and fluid behavior.
Both concepts are widely used in science, engineering, and daily life applications. By clearly understanding the difference between mass and density, students can build a strong foundation in physics and better interpret the physical world around them. This knowledge also helps in solving real-life problems involving materials, motion, and environmental science.

SwiftHarbor is a dedicated English professor, language researcher, and the founder of SpellCompare.com. With years of academic experience in English grammar, vocabulary development, and linguistic comparison, SwiftHarbor specializes in simplifying complex language rules into clear, practical explanations.
As an expert in word usage, spelling differences, and commonly confused terms, SwiftHarbor has helped thousands of learners improve their writing accuracy and communication skills. Through SpellCompare.com, he provides detailed comparisons, easy examples, and research-based insights that make English learning accessible for students, writers, and professionals worldwide.
Known for a clear teaching style and structured explanations, SwiftHarbor focuses on eliminating confusion between similar words, improving grammar confidence, and promoting precise communication. His mission is simple: to make English easier, clearer, and more powerful for everyone.