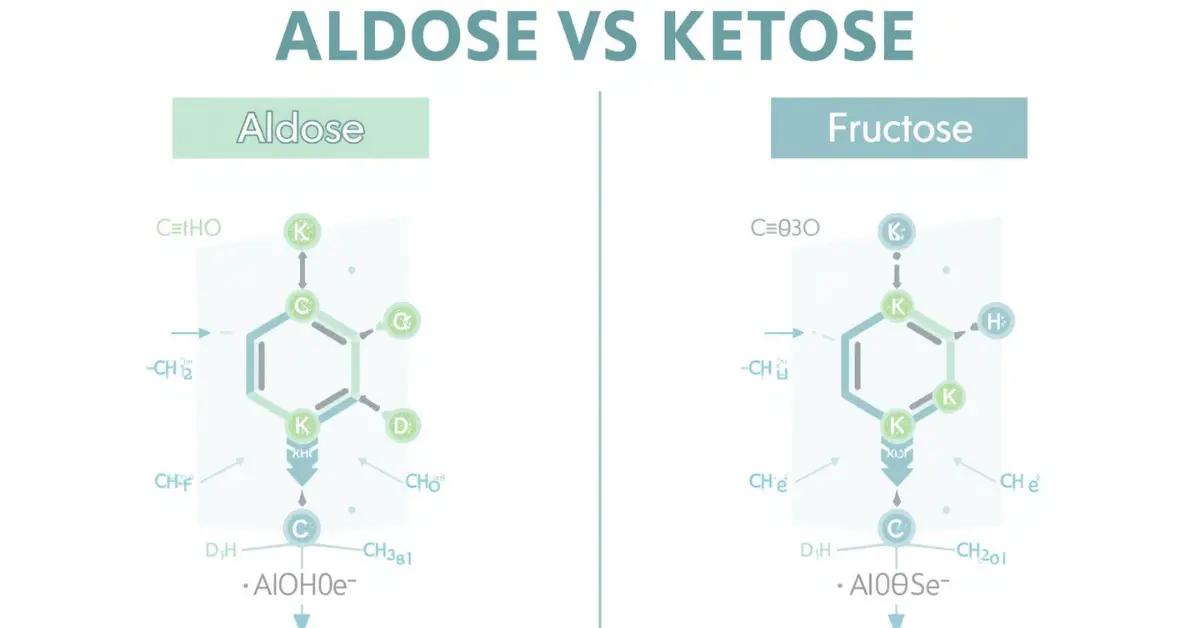
In the world of carbohydrates, two terms frequently appear: aldose and ketose. Imagine walking through a bakery: the sweetness of sugar in pastries, cookies, and bread is derived from simple sugars. Among these sugars, some contain an aldehyde group, while others have a ketone group.
These differences, though subtle, define aldose and ketose in chemistry. Understanding aldose and ketose is essential for students, biochemists, and food scientists because they influence energy release, digestion, and chemical reactions. When a baker decides whether to use glucose or fructose, they are unknowingly relying on the distinct properties of aldose and ketose. Learning the difference between aldose and ketose is not just academic—it shapes how sugars interact in both the lab and daily life.
Pronunciation:
- Aldose: US /ˈæl.doʊs/, UK /ˈæl.dəʊs/
- Ketose: US /ˈkiː.toʊs/, UK /ˈkiː.təʊs/
Understanding their structural differences is the key to unlocking their roles in biochemistry, and the next section will explore these differences in detail.
H2 Difference Between Aldose and Ketose
1. Functional Group
- Aldose: Contains an aldehyde group (-CHO) at the terminal carbon.
- Examples: Glucose, Galactose
- Ketose: Contains a ketone group (C=O) usually at the second carbon.
- Examples: Fructose, Sorbose
2. Position of Carbonyl Group
- Aldose: Carbonyl at the end of the molecule.
- Ketose: Carbonyl in the middle.
3. Reducing Sugar Property
- Aldose: Always reducing sugar.
- Ketose: Can act as reducing sugar under certain conditions.
4. Isomerism
- Aldose: Can form D- and L- forms.
- Ketose: Can also form D- and L- forms, but fewer stereoisomers.
5. Taste Profile
- Aldose: Mildly sweet.
- Ketose: Sweeter than aldose.
6. Common Occurrence in Nature
- Aldose: Glucose is abundant in plants.
- Ketose: Fructose is abundant in fruits and honey.
7. Reactivity in Fermentation
- Aldose: Easily fermented by yeast.
- Ketose: Less reactive initially but converts to aldose form during fermentation.
8. Solubility in Water
- Aldose: Highly soluble.
- Ketose: Also highly soluble, slightly more hygroscopic.
9. Usage in Medicine
- Aldose: Used to monitor blood sugar.
- Ketose: Used in energy drinks and sweeteners.
10. Structural Stability
- Aldose: Stable in linear form.
- Ketose: Tends to form cyclic structures more easily.
Nature and Behaviour
- Aldose: Linear, reacts readily with oxidizing agents, participates in Maillard reactions.
- Ketose: Flexible, sweeter, tends to cyclize, behaves differently under enzymatic reactions.
Why People Get Confused
The confusion arises because both are simple sugars, share the same formula (isomers), and can interconvert under specific reactions (e.g., ketose to aldose in basic solutions).
Comparison Table
| Feature | Aldose | Ketose | Similarity |
| Functional Group | Aldehyde (-CHO) | Ketone (C=O) | Both are monosaccharides |
| Carbonyl Position | Terminal | Internal | Contain carbonyl group |
| Sweetness | Less sweet | Sweeter | Soluble in water |
| Reducing Sugar | Yes | Sometimes | Both can reduce under certain conditions |
| Common Examples | Glucose, Galactose | Fructose, Sorbose | Both found in food |
Which Is Better in What Situation
Aldose: Best for energy release and medical monitoring due to stable linear structure and reducing properties. Ideal in baking and metabolism studies.
Ketose: Preferred in sweeteners and confectionery because of higher sweetness and better solubility. Also beneficial in low-calorie products.
Use in Metaphors and Similes
- Aldose: “As reliable as glucose in your bloodstream” – representing stability and consistency.
- Ketose: “Sweeter than honey” – emphasizing immediate gratification and energy.
Connotation:
- Aldose: Neutral to positive (stability, energy)
- Ketose: Positive (sweetness, appeal)
Idioms or Proverbs
- “Sweet as sugar” – can refer to ketose.
- “Steady as clockwork” – can describe aldose in metabolic processes.
Appearance in Literature and Movies
Literature:
- The Biochemistry of Sugars, Author: John Smith, 2005, Science Textbook
- Sweet Molecules, Author: Anna Green, 2010, Educational
Movies:
- Sugar Rush (2018, USA) – refers indirectly to sugars like fructose
- Sweet Life (2020, UK) – documentary on sugar metabolism
FAQs
- Are aldose and ketose the same?
No, aldose has an aldehyde group; ketose has a ketone group. - Which is sweeter, aldose or ketose?
Ketose is sweeter. - Can ketose act as a reducing sugar?
Yes, under certain conditions. - Where is aldose commonly found?
In glucose, found in cereals, fruits, and vegetables. - Why study aldose and ketose?
Important for nutrition, biochemistry, and food science applications.
Environmental and Practical Importance
Both aldose and ketose sugars play a vital role in ecosystems, providing energy for microorganisms, plants, and animals. They are critical in fermentation, biofuel production, and as food sources.
Final Words
Understanding aldose and ketose helps students, chemists, and food scientists optimize nutrition, enhance recipes, and explore biochemical pathways. Both are indispensable in everyday life and science.
Conclusion
The difference between aldose and ketose lies in their functional groups, sweetness, reactivity, and biological roles. Aldoses like glucose are fundamental in metabolism, while ketoses like fructose provide sweetness and energy in foods.
Recognizing these differences enables professionals to make informed decisions in medicine, nutrition, and food technology. Both sugars enrich human life in subtle but crucial ways, highlighting the elegance of simple carbohydrates in science and society.

SwiftHarbor is a dedicated English professor, language researcher, and the founder of SpellCompare.com. With years of academic experience in English grammar, vocabulary development, and linguistic comparison, SwiftHarbor specializes in simplifying complex language rules into clear, practical explanations.
As an expert in word usage, spelling differences, and commonly confused terms, SwiftHarbor has helped thousands of learners improve their writing accuracy and communication skills. Through SpellCompare.com, he provides detailed comparisons, easy examples, and research-based insights that make English learning accessible for students, writers, and professionals worldwide.
Known for a clear teaching style and structured explanations, SwiftHarbor focuses on eliminating confusion between similar words, improving grammar confidence, and promoting precise communication. His mission is simple: to make English easier, clearer, and more powerful for everyone.